Laurent J. Bouyer1,2, François Routhier1,2, Caroline Rahn1, Bradford J. McFadyen1,2
1Center for interdisciplinary research in rehabilitation and social integration, Institut de réadaptation en déficience physique de Québec, Québec, Canada; 2Department of Rehabilitation, Université Laval, Québec, Canada.
Abstract
Three individuals with Multiple Sclerosis (MS) were tested in a laboratory setting while wearing a mobility-assisting device called a dermoskeleton. The tested device used was the B-TEMIA’s military-based version dedicated to military applications, with the necessary design adjustments for the proposed study. Participants performed standardized tasks (Time Up and Go, Stair Test, 6 Minute Walk Test) with and without the dermoskeleton, and execution times were compared. Pilot data show improvement of gait with the device in 2 of the 3 participants, in terms of stability (higher inter-step hip kinematics coherence) and endurance (longer distance during the 6 Minute Walk Test). The dermoskeleton was moderately to highly valued by participants during the laboratory tests. While the results from this pilot study suggest a potential for dermoskeletal technology in improving mobility in persons with MS, additional tests in a community setting are now required to quantify the efficacy and safety of the innovation as well as how the tested device can be used in daily activities, and more broadly improve the quality of life of its users.
background
B-TEMIA Inc. (www.b-temia.com) develops and markets “dermoskeletons” to enhance and augment mobility through biomechanical assistance using motorized orthopaedic supports controlled by computers. At the core of their system is the integration of three components: 1) a network to sense the biomechanical characteristics of a specific user; 2) software for movement recognition to characterize gait phases and movement status; and 3) software to control the dermoskeleton joint mechanism to optimize biomechanical assistance. Contrary to classical exoskeleton technology, dermoskeletons are not self weight bearing. They use the person’s musculoskeletal system as the base of support and a means of transferring force to the joints. They are therefore smaller and lighter than their exoskeletal counterparts.
In the past, B-TEMIA’s dermoskeletal technology has been used successfully with military personnel to enhance endurance and weight bearing in operational situations. Due to its capacity to augment force generation during mobility tasks, the general goal of the present study was to evaluate in a pilot study if such technology could also be useful as a means to improve endurance in persons with muscle weakness due to central nervous system lesions.
Individuals with multiple sclerosis (MS) were chosen as a key population to address initial questions regarding the rehabilitation potential of the dermoskeletal technology. MS is an unpredictable, disabling disease of the central nervous system (Scalfari, Neuhaus, Degenhardt, Rice, Muraro, Daumer & Ebers, 2010; Yildiz, 2012). Gait disturbances (difficulty in walking) in MS are not unusual and several factors may be involved. Muscle weakness, increased tone and spasticity, balance problems, coordination problems and even fatigue and pain can all play a role. As gait is controlled by neural pathways distributed across the central nervous system (Grey, Bouyer & Nielsen, 2012), the disseminated nature of MS makes it possible for many important nerve tracts to be disrupted by the disease. Dermoskeletal technology has the potential to provide the assistance in walking needed by this population to increase endurance and to promote better and safer gait in activities of daily living that are compromised by MS. It could also delay the need for home modifications, or of becoming permanently wheelchair bound.
objectiveS
The main objective of this study was to describe the potential effects of dermoskeletal technology on gait and endurance during mobility tasks of individuals with MS. A secondary objective was to describe the perception of potential users regarding this technology.
Method
Design
An experimental single-subject design was adopted.
Dermoskeleton fitting
A modified version of B-TEMIA’s military dermoskeleton was used to test participants. Prior to each data collection, it was adjusted to each participant’s anthropometric characteristics. Proper fitting is very important to obtain an appropriate and efficient man-machine interface.
Participants
Initial screening for participants was carried out using the iMed database of the Institut de réadaptation en déficience physique de Québec (IRDPQ, Québec, Canada) as well as thorough physiotherapy chart review. Four individuals with MS were initially targeted and underwent a clinical examination by a physiotherapist familiar with MS to characterize their impairments, walking capacity, lower limb muscle strength, knee flexion and extension mobility (range of motion), maximum active and passive ankle dorsiflexion angle, lower limb spasticity, lower limb light touch perception, standing static balance, standing dynamic balance, comfortable walking speed and participant’s ability to ascend and descend stairs was assessed. This examination allowed the identification of participants potentially able to complete the protocol and to provide information for the secondary objective of this study. In order to fit the dermoskeleton technology, all patients had to measure between 168 cm and 188 cm. They had to be free of any neurological conditions other than MS (Parkinson’s disease, stroke, etc), no ataxic gait, unstable cardiovascular condition, pulmonary disease, or cognitive/communication impairments that could affect their participation in the study. Finally, they had to be diagnosed with primary progressive or secondary progressive MS, with an Expanded Disability Status Scale (EDSS) between 4.0 and 6.5 (moderate to severe difficulty with ambulation) and to be medically stable.
Data collection and measurements
Each participant was met five times within a period of 1 month. Visit 1 involved taking anthropometric measures and visually assessing their unassisted gait pattern. Following this visit, B-TEMIA optimized the dermoskeleton’s contact elements for a better fit with the participants’ specific morphology. Initial software adjustments were also made to take into account the more hesitant pathological gait patterns. At visit 2, participants performed a series of 3 standardized tasks without the dermoskeleton to establish baseline performance (Timed Up and Go [TUG], Modified Timed Stair Test [MTST], and the 6 Minute Walk Test [6MWT] – see next subsection for details). During all three tasks, muscle activity (EMG) was recorded bilaterally from representative muscles of the lower extremities using a wireless EMG amplifier (Noraxon 2400T, Scottsdale, USA), and knee and hip flexion/extension angles were recorded at the joints of the most affected lower extremity using electrogoniometers (Biometrics Ltd, Ladysmith, USA). Electrode placement was determined using Surface ElectroMyoGraphy for the Non-Invasive Assessment of Muscles (SENIAM) project recommendations. Muscles recorded from were: Rectus Femoris (RF; hip flexor/knee extensor), Vastus Lateralis (VL; knee extensor), Medial Gastrocnemius (MG; knee flexor, ankle plantarflexor), Medial Hamstring (MH; hip extensor/knee flexor) and Tibialis Anterior (TA; ankle dorsiflexor). EMG signals were amplified and band pass filtered (30-450 Hz). At visit 3, participants were fitted and familiarized with the modified dermoskeleton; no data was collected. Visit 4 was a second familiarization session where participants experimented walking and moving around with the dermoskeletal technology prior to performing the standardized tasks. This session was necessary to allow the participant to become accustom to the system and its control. Control parameters (targeted gait phases; required torque amplitudes; optimal torque profiles) were adjusted manually during this visit to maximize performance on the standardized tasks. Finally, at visit 5, the participants repeated the 3 standardized tasks performed at visit 2, this time with the dermoskeleton in both passive and assistive modes (except for the 6MWT, that was only performed in assistive mode to prevent exhaustion). Participants did not know the control mode for the dermoskeleton. They were simply told to perform the tasks twice. The passive mode was used to control for any placebo effect. Measures of change in performance (visits 2 vs 5) were obtained using the single-subject design.
A semi-structured interview (20 questions) consisting of questions regarding the participants’ habits (walking and general activity level) as well as their initial impression of the dermoskeleton was carried out in person immediately following visit 5. A brief follow up questionnaire was also administered by phone the next day, to verify if any adverse effects were felt and if the participants had any other questions regarding the device.
Tasks description
The first task selected was the TUG, a timed test that assesses functional mobility: rising from a chair, walking three meters, pivoting, walking back to the chair and siting down. The second task selected was the MTST: time to ascend turn and descend a 5 steps starting from a standing position. The MTST was modified from the original TST for safety considerations, functional significance and its lower impact on MS participants’ fatigue. The third task selected was the 6MWT, a measure of walking endurance: total distance covered by a participant asked to walk as fast and far as possible for 6 minutes. In addition to the standardized tasks, participants were asked to rate their level of exertion before and after the 6 MWT using the Borg Rate of Perceived Exertion (RPE) Scale: linear scale ranging from 6 (no feeling of exertion) to 20 (very, very hard).
Analysis
For each of the 3 standardized tasks, execution times without and with the dermoskeleton were compared (chronometric data). In addition, for the 6 MWT, stride-to-stride movement consistency within a session was quantified using cross correlations on hip angular displacement data. Qualitative data from the semi-structured interviews were summarized and grouped by theme for each question, and then descriptively analyzed.
Results AND DISCUSSION
Three of the 4 selected individuals with MS completed testing (3 males, age range 49-65 yrs). Drop out by the fourth participant was due to improper fitting of the dermoskeleton causing slippage of the unit (participant #4; female). Only data from the 3 participants are reported and analyzed.
Chronometric/distance data
Chronometric/distance data can be found in Table 1 below. For the 6MWT test, fitting and tuning were optimal in participant #3 and as a result, a 57 m increase in walking distance was measured which is close to a clinically meaningful improvement for this patient population which is situated between 60-90 m. For the other participants (#1 and #2), there were difficulties in tailoring the control algorithm for the needs of each individual. Participant #2 could not complete the 6MWT test due to inappropriate timing of the drive system. Moreover, it can be seen that for TUG and MTST, the dermoskeleton did not negatively impact performance and the task times with the dermoskeleton remained similar in range. It is expected that with proper software tuning, improvement in performance may be possible in the future.
With Dermoskeleton |
|||||
Task |
Participant |
Baseline |
No assist |
Assist |
Difference |
TUG (s) |
1 |
14.44 |
13.93 |
13.99 |
0.45 |
2 |
15.48 |
16.98 |
16.01 |
-0.53 |
|
|
3 |
14.69 |
14.85 |
13.32 |
1.37 |
MTST (s) |
1 |
15.39 |
15.37 |
16.28 |
-0.89 |
2 |
12.36 |
16.18 |
16.22 |
-3.86 |
|
|
3 |
15.89 |
13.41 |
13.78 |
2.11 |
6MWT distance (m) |
1 |
365 |
n/a |
315 |
50 |
2 |
210 |
n/a |
not completed |
n/a |
|
|
3 |
329 |
n/a |
386 |
-57 |
6MWT (# of steps) |
1 |
512 |
n/a |
506 |
6 |
|
2 |
450 |
n/a |
134* |
316 |
|
3 |
510 |
n/a |
550 |
-40 |
| *: Not completed | |||||
Electromyography
While good care was taken in collecting surface electromyography (EMG) from the participants with and without the dermoskeleton, placing he EMG electrodes underneath some of the padding elements of the device produced significant motion artefacts in the recorded signals. While pre-processing of the signals allowed removal of some motion artefacts, the remaining artefacts were sufficiently large to prevent a detailed analysis of the muscle activation profiles. Based on this finding and on a review of other types of surface EMG electrodes, we thus recommend that future work uses flat surface electrodes rather than snap-lead interfaces. This will reduce electrode thickness, and hence the mechanical interference of the dermoskeleton on the recorded signals.
Movement pattern
While EMG signals could not be used, electrogoniometer data allowed us to quantify how movement patterns were affected by the dermoskeleton. Indeed, while participant #3 improved his walking distance by 57 meters as noted above, it is important to also know if movement quality with the dermoskeleton was maintained, or if the device moved the participant in a non-physiological manner. The latter would be problematic in the long-term, as it could cause “abnormal use injury” to the locomotor apparatus, and thus produce more harm than benefits to the potential end-user.
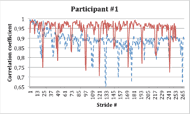
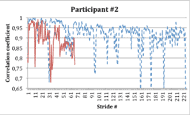
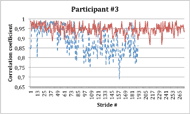 Figure 1: Movement consistency for participants #1 to #3 –Correlation to first 10 strides (red: With dermo; blue/dashed: Without dermo).
Figure 1: Movement consistency for participants #1 to #3 –Correlation to first 10 strides (red: With dermo; blue/dashed: Without dermo).While measuring actual forces transferred to the individual joints was beyond the scope of the present study, movement consistency over time, a measure of the quality of movement, was assessed by comparing the shape of hip movement trajectories from one step to the next using a cross correlation function. The cross correlation compares a given movement trajectory to a template (in this case computed from the mean of the first 10 gait cycles). If the given trajectory is identical to the template, then the cross correlation will give a value of 1. If the two do not relate, the value will drop to 0. Figure 1 (bottom) presents the cross correlation value for every gait cycle for participant #3 without (blue/dashed trace) or with the dermoskeleton in assist mode (red trace). It can be seen that in the absence of the dermoskeleton, his gait starts to degrade after 60 strides. This can be interpreted as a sign of fatigue or loss of optimal movement control. When wearing the dermoskeleton, the cross-correlation remains high for the entire duration of the test, showing that even when fatigue begins, the device can maintain a good movement consistency. This is likely part of the reason why the participant was able to walk a longer distance with the dermoskeleton.
The inverse can be seen in participant #2 (Figure 1 middle), where coherence was reduced in the presence of the dermoskeleton. This was associated with the participant stopping before the 6MWT was complete. Finally, participant #1 (Figure 1 top), who had a relatively good fit, showed a higher cross correlation coefficient with the dermoskeleton, similar to participant #3.
Interviews
Based on the semi-structured interviews data, some interesting information can be extracted. Overall, the prototype dermoskeleton was moderately to highly valued by participants during laboratory tests. They felt it’s possible to use this technology in their daily activities that require lots of energy or stability such as: grocery shopping, mowing the lawn, etc. One of the participants said that he felt more normal and natural while wearing the device. However participants expressed that the design of the B-TEMIA’s clinical version should include specific adjustments such as ligher weight and user-friendly attachments to better address the user requirements of the MS population. Also, participants would like to test over a longer period in the home environment in the home environment. Participants see some potential in this technical assistance for locomotion. Finally, all the three particpants felt the assistive force when wearing the dermoskeleton.
conclusion
We succeeded our initial evaluation of the potential effects of the B-TEMIA’s dermoskeletal technology on the mobility of individuals with MS, in 3 of the 4 recruited participants (objective 1). Although our sample size is small, our preliminary results suggest that the dermoskeletal technology may benefit persons with MS, when properly adjusted for fitting and control. In the future, additional studies may be conducted with other populations in order to define the full potential and limits of this technology in a larger spectrum of individuals with disabilities. Recommended laboratory studies could include a detailed biomechanical analysis of assistive force distribution to the lower limb joints during gait with the dermoskeleton, to improve understanding of the interaction with the human body. Finally, studies in community settings are also needed to validate how this new assistive device can impact the user’s participation in daily activities and quality of life.
References
Grey, M.J., Bouyer, L., Nielsen, J.B. (2012). Chapter 10 - Neural control of walking. In Motor Control And Motor Learning. Publisher: Routledge. Editors: Gollhofer A, Taube W, Nielsen JB. pp. 197-212.
Scalfari, A., Neuhaus, A., Degenhardt, A., Rice, G.P., Muraro, P.A., Daumer, M., Ebers, G.C. (2010). The natural history of multiple sclerosis: a geographically based study. 10. Relapses an long-term disability. Brain, 133, 1914-1929.
Yildiz, M. (2012). The impact of slower walking speed on activities of daily living in patients with multiple sclerosis. International Journal of Clinical Practice, 66, 11, 1088-1094.
aknowledgements
This project was funded by the Natural Sciences and Engineering Research Council (NSERC) of Canada through a partnership Engage Grant. Authors would also like to thank B-TEMIA Inc. (Québec, Canada) and the Multiple Sclerosis and Neuromuscular Disease Program of the Institut de réadaptation en déficience physique de Québec (Québec, Canada) for their close collaboration.
Audio Version PDF Version