Nathan S. Hogaboom1,2, Michelle L. Oyster1,3, Alicia M. Koontz1,2, Michael L. Boninger1,3
1. Human Engineering Research Laboratories, VA Pittsburgh Healthcare System, Pittsburgh, PA
2. Department of Rehabilitation Science and Technology, University of Pittsburgh, Pittsburgh, PA
3. Department of Physical Medicine and Rehabilitation, University of Pittsburgh, Pittsburgh, PAABSTRACT
Twenty-one wheelchair users with spinal cord injuries (SCI) were recruited for this cross-sectional, repeated-measures study. The objectives were to investigate ultrasonographic changes in median nerve size after repeated transfers, and identify if proper hand placement influences these changes. Median nerve cross-sectional area and swelling ratio – ultrasonographic markers for carpal tunnel syndrome (CTS) – were measured before and after transfers. Participants who used hand placement recommended by Clinical Practice Guidelines had less median nerve changes. Results from this study indicate transfers cause median nerve changes associated with CTS, and improper hand placement can exacerbate these changes. The small, heterogenous sample makes it difficult to generalize to the entire SCI population. However, results warrant further investigation into the efficacy of transfer training interventions in reducing risk of developing CTS.
INTRODUCTION
Approximately 273,000 individuals with spinal cord injuries (SCI) live in the United States (NSCISC, 2013), the majority of whom use a wheelchair for mobility. Among these individuals, carpal tunnel syndrome (CTS) is one of the most common causes of upper extremity pain (Boninger, et al. 2005). CTS is characterized by chronic compression of the median nerve within the carpal tunnel that results in pain, numbness, tingling, and weakness. As manual wheelchair users with SCI rely on the use of their upper extremities to perform activities of daily living, hand and wrist pain can negatively affect quality of life and functional independence. Development of CTS has been attributed to performance of repetitive wheelchair activities, such as transfers, that impose high forces on the wrist while in awkward positions. Clinical Practice Guidelines recommend certain hand placements to help redistribute wrist forces in a manner that reduces stress on the median nerve (Figure 1; Boninger, et al. 2005).
Ultrasound provides investigators and clinicians with a non-invasive, low-cost, method to measure acute changes in soft tissue. A quantitative ultrasound (QUS) imaging protocol was developed to reliably measure changes in ultrasonographic parameters indicative of CTS (Impink, Gagnon, Collinger, & Boninger, 2010). This technique was used to identify acute changes in median nerve characteristics after wheelchair propulsion (Impink, Collinger, & Boninger, 2011). Using the same method, this study aimed to measure changes in median nerve characteristics after wheelchair transfers.
It was hypothesized that repeated transfers would result in changes in ultrasonographic measures of median nerve health, and participants who did not utilize proper hand placement during the transfer would experience greater changes than those who did.
METHODS
Subjects
Participants were recruited through research registries, flyers, word of mouth, and at the 2013 National Veterans Wheelchair Games. Participants were included in the study if they were greater than 18 years old, had a non-progressive SCI that occurred over one year prior to participation in the study, used a wheelchair for over 40 hours/week, and were able to independently transfer to and from a surface within 30 seconds. They were excluded if they had any arm pain that limited their ability to transfer, actively used their leg muscles when transferring, or had a current or recent history of pressure sores or cardiopulmonary issues. Institutional Review Board approval was obtained prior to implementation of study procedures. All participants provided written informed consent.
Baseline Questionnaires and Examinations
Participants provided demographic information and completed the Boston Carpal Tunnel Questionnaire (BCTQ; Levine, et al. 1993) to assess baseline levels of wrist pain. Participants underwent a physical exam evaluating upper and lower extremity motor scores according to the International Standards of Neurological Classification of SCI (Kirshblum, et al., 2011).
Quantitative Ultrasound Examination
A QUS exam of the median nerve was completed wherein images of the non-dominant wrist were collected using a previously described technique (Impink, Gagnon, Collinger, & Boninger, 2010). Participants were positioned to sit upright in their own wheelchairs with their elbows flexed to 90 degrees and forearms supinated. Wrists and fingers were relaxed and resting on the ipsilateral thigh. Images were collected at the pisiform and distal radius by positioning the probe in the transverse plane and perpendicular to the palmar side of the wrist. To image the level of the radius, the probe was dragged distally until the edge of the radius dropped out of view, then proximally until it reappeared. The pisiform level was found by moving the probe distally until the pisiform appeared in the image.
Depth and focus were held constant between participants. A single investigator collected and analyzed all images; this technique has high reliability when images are collected and analyzed by the same investigator (Impink, Gagnon, Collinger, & Boninger, 2010). The non-dominant wrist was chosen to control for nerve changes that may be associated with activities performed by the dominant wrist other than transfers (e.g. writing).
Transfer Protocol
All transfers were performed using the participants’ own wheelchairs. Transfer quality was graded by two investigators using the reliable Transfer Assessment Instrument (TAI; Tsai, Rice, Hoelmer, Boninger, & Koontz, 2013). Four transfers were graded using the TAI under two conditions: to-and-from a surface level to their wheelchair seats and to-and-from a surface two inches higher than their wheelchair seats. The TAI is a two-part evaluation. Part one assigns fifteen items either a “yes” or “no” depending on whether they met the item criteria; part two assigns eight items a score from 0 to 4 based on the degree to which criteria were met. Items include wheelchair placement, trunk and shoulder movement, and hand placement (Tsai, Rice, Hoelmer, Boninger, & Koontz, 2013).
Item 10 from Part 1 was used in the present analysis. This item qualifies whether participants placed the hand of their trailing arm in a position recommended by Clinical Practice Guidelines (Boninger, et al. 2005) during the transfer. Recommended hand placement is described as gripping the edge of a surface within the base of support (e.g. wheelchair armrest, edge of mat table or bed, etc.; Figure 1A). If the individual has insufficient motor strength to grip a surface the palm should be flat on the surface with flexed fingers to maintain a tenodesis grasp (Figure 1B). The trailing arm was of interest as it experiences the greatest vertical reaction forces during transfers (Gagnon, Nadeau, Noreau, Dehail, & Gravel, 2008).
After completing the four transfers that were graded using the TAI, participants completed eighteen transfers to and from a mat table of varying heights; this number is based on the estimated number of transfers completed per day by people with SCI (Samuelsson, Tropp, & Gerdle, 2004). The first and last set of six transfers was onto a level surface, while the middle set of six was onto a surface two inches higher than their wheelchair seats. A 60-second break was given between each set to provide rest. Subjects completed one transfer per fifteen seconds, which was controlled with a metronome. At the end of the transfer protocol, participants rated their perceived level of exertion using a Borg scale.
Image Analysis
Median nerve variables of interest were cross-sectional area (CSA) at the pisiform and swelling ratio (SR). These are two ultrasound parameters highlighted in the literature as having the highest specificity and sensitivity for diagnosis of CTS (Beekman & Visser, 2003). A MATLAB program was used to analyze all images in a random order while the investigator was blinded from time point and subject. CSA was determined by boundary trace along the circumference of the nerve and reflects its two-dimensional size on the image; SR was calculated by dividing CSA at the pisiform by CSA at the radius. Complete details of the image analysis protocol have been published previously (Impink, Gagnon, Collinger, & Boninger, 2010).
Statistical Analysis
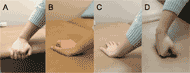 Figure 1: Hand placement when transferring to a wheelchair – recommended (A, B) and non-recommended (C, D).
Figure 1: Hand placement when transferring to a wheelchair – recommended (A, B) and non-recommended (C, D).
Significance was set a priori to p<.05. Data were assessed for normality using the Kolmogorov-Smirnov test. Participants were dichotomized into groups based on whether or not they utilized recommended hand posture during transfers. Between-groups differences at baseline were assessed using Fisher’s exact test (categorical), Mann-Whitney U tests (discontinuous), and independent t-tests (continuous). Both hypotheses were tested using two-way mixed-model analyses of variance. Independent variables included the intervention (repeated transfers) and group (hand placement). Dependent variables included median nerve CSA at the pisiform and SR. A Bonferroni correction was applied to account for multiple tests run on the two QUS variables (p<.05/2=.025).
RESULTS
Subjects
A convenience sample of 32 participants was recruited for the study; data from 21 were included in the final analysis. Wrists of the eight subjects with tetraplegia were not imaged as differences in motor control may alter median nerve responses to activity and confound results. Three participants used their leg muscles when transferring and thus their data were not analyzed. Demographic variables (Table 1) were not different between groups.
|
Improper (N = 6) | Proper (N = 15) | Total Group |
|---|---|---|---|
Age (years) |
48.8 (15.1) |
41.7 (10.8) |
43.7 (12.2) |
Years Since Injury |
16.6 (13.5) |
14.2 (9.2) |
14.9 (10.3) |
Weight (kg) |
79.2 (32.1) |
78.0 (19.7) |
78.3 (23.0) |
ASIA – UL |
50 |
49 |
50 |
BCTQ |
1.03 |
1.32 |
1.05 |
Borg |
6.5 |
7 |
7 |
Gender |
5 Male 1 Female |
13 Male 2 Female |
|
Race/ Ethnicity |
1 Black 4 White 1 Hispanic |
7 Black 6 White 1 Hispanic 1 Multiracial |
|
Analyses of Variance
There was a significant main effect of repeated transfers on CSA and SR (Table 2). No main effect of hand position was observed on CSA or SR. An interaction effect was observed between hand position and repeated transfers on SR (F(1,19)=6.420, p=.020), but not CSA (F(1,19)=2.753, p=.113). Interaction effects on SR and CSA are graphically presented in Figures 2 and 3.
DISCUSSION
Median nerves of participants in the current study experienced acute increases in size as a result of repeated transfers. Median nerves became larger at the level of the pisiform – the location that corresponds to the inlet of the carpal tunnel – which is a response associated with CTS. During a transfer, most of the body weight is supported by the upper limbs. Large vertical forces are experienced by both arms during the lift phase, more so by the trailing arm (Gagnon, Nadeau, Noreau, Dehail, & Gravel, 2008). The hand serves as the interface between person and surface, so it is likely the wrist experiences a percentage of these forces which act to compress the carpal tunnel or put tension on the tendons and median nerve within. Transfers are necessary for functional independence so it is important to understand the mechanisms behind the development of CTS and how transfers contribute.
N = 21 |
Baseline |
Post-Transfers |
F |
df |
p |
|---|---|---|---|---|---|
CSA (mm2) |
11.86 (2.43) |
12.23 (2.69) |
6.121 |
1, 19 |
.023 |
SR |
0.99 (0.14) |
1.01 (0.14) |
6.258 |
1, 19 |
.022 |
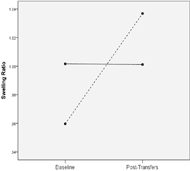 Figure 2: Interaction effect of transfers and hand placement on median nerve swelling ratio. Dotted line = non-recommended hand placement; solid = recommended.
Figure 2: Interaction effect of transfers and hand placement on median nerve swelling ratio. Dotted line = non-recommended hand placement; solid = recommended.Participants who placed their trailing hands in a position recommended by Clinical Practice Guidelines (Figures 1A-D; Boninger, et al. 2005) experienced lesser changes than those who did not. This position encourages a more neutral wrist and prevents extreme wrist and finger extension, which can increase intra-carpal canal pressure and median nerve displacement under the transverse carpal ligament (Werner & Andary, 2002). Repeated exposure to large forces, particularly while the wrists and fingers are extended, could impose both mechanical and ischemic damage on the nerve. Ultimately, damage can accumulate and lead to conduction block, axonal death, and myelin degradation (Werner & Andary, 2002). These underlying physiological mechanisms result in symptoms commonly associated with CTS. This study provides further evidence that teaching individuals to utilize recommended hand position while transferring will diminish their risk of developing CTS.
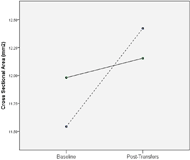 Figure 3: Interaction effect of transfers and hand placement on median nerve cross-sectional area, in millimeters squared. Dotted line = non-recommended hand placement; solid = recommended.
Figure 3: Interaction effect of transfers and hand placement on median nerve cross-sectional area, in millimeters squared. Dotted line = non-recommended hand placement; solid = recommended. There were a few limitations to this study. The sample was relatively small and heterogenous with respect to gender, with uneven group sizes based on hand placement. These limitations could affect the power of statistical tests and hamper generalizability to the SCI population. Transfer direction was not included in the analysis and may influence median nerve changes in addition to hand placement.
CONCLUSIONS
Repeated wheelchair transfers induced acute ultrasonographic changes to the median nerve associated with CTS. Individuals who placed the trailing hand in a recommended position did not demonstrate these changes. Further investigation is warranted to determine the efficacy of transfer training interventions in reducing risk of developing CTS.
REFERENCES
Beekman, R., & Visser, L.H. (2003). Sonography in the diagnosis of Carpal Tunnel Syndrome: A critical review of the literature. Muscle and Nerve, 27, 26-33.
Boninger ML, Waters RL, Chase T, Dijkers MPJM, Gellman H, Gironda RJ. (2005) Preservation of upper limb function following spinal cord injury: A clinical practice guideline for health-care professionals. Journal of Spinal Cord Medicine, 28, 434-470.
Gagnon, D., Nadeau, S., Noreau, L., Dehail, P., & Gravel, D. (2008). Quantification of reaction forces during sitting pivot transfers performed by individuals with spinal cord injury. Journal of Rehabilitation Medicine, 40, 468-476.
Impink, B.G., Gagnon, D., Collinger, J.L., Boninger, M.L. (2010). Repeatability of ultrasonographic median nerve measures. Muscle and Nerve, 41, 767-773.
Impink, B.G., Collinger, J.L., Boninger, M.L. (2011). The effect of symptoms of Carpal Tunnel Syndrome on ultrasonographic median nerve measures before and after wheelchair propulsion. PM&R, 3, 803-810.
Kirshblum, S.C., Burns, S.P., Biering-Sorensen, F., Donovan, W., Graves, D.E., Jha, A., … & Waring, W. (2011). International standards for the neurological classification of Spinal Cord Injury (Revised 2011). The Journal of Spinal Cord Medicine, 34, 535-546.
Levine, D.W., Simmons, B.P. Koris, M.J., Daltroy, L.H., Hohl, G.G., Fossel, A.H., & Katz, J.N. (1993). A self-administered questionnaire for the assessment of severity of symptoms and functional status in carpal tunnel syndrome. Journal of Bone and Joint Surgery-American Volume, 75,1585-1592.
National Spinal Cord Injury Statistical Center. (2013). Spinal cord injury facts and figures at a glance. Journal of Spinal Cord Medicine, 36, 170-171.
Samuelsson, K.A.M., Tropp, H., & Gerdle, B. (2004). Shoulder pain and its consequences in paraplegic spinal cord-injured, wheelchair users. Spinal Cord, 42, 41-46.
Tsai, C.Y., Rice, L.A., Hoelmer, C., Boninger, M.L., Koontz, A.M. (2013). Basic psychometric properties of the Transfer Assessment Instrument (version 3.0). Archives of Physical Medicine and Rehabilitation, 94, 2456-2464.
Werner, R., & Andary, M. (2002). Carpal tunnel syndrome: Pathophysiology and clinical neurophysiology. Journal of Clinical Neurophysiology, 113, 1373-1381.
ACKNOWLEDGEMENTS
The contents of this paper do not represent the views of the Department of Veterans Affairs or the United States Government. This project was supported by the National Institute on Disability and Rehabilitation Research, Office of Special Education and Rehabilitation Services, U.S. Department of Education (H133N110011). This material is based upon work supported by the National Science Foundation Graduate Research Fellowship (Grant #1247842). Any opinion, findings, and conclusions or recommendations expressed in this material are those of the authors and do not necessarily reflect the view of the National Science Foundation.
Audio Version PDF Version