Motor-activated Multi-functional Wrist Orthotic to Assist Individuals with Cervical Spinal Cord Injuries with Activities of Daily Living
Shruthi Suresh1, Daniel F. Madrinan Chiquito2, Sudhanshu Manda1, Lucas Jacob3, Bradley S. Duerstock1,4
Weldon School of Biomedical Engineering1, National University of Colombia, Palmira2, Purdue Polytechnic Institute3, School of Industrial Engineering4, Purdue University
Abstract
Many individuals with mid-cervical spinal cord injuries (SCI) rely on wrist supports or orthotics in order to keep their wrists stabilized to grant them more functional ability. However commercially-available wrist supports principally provide support to perform limited activities of daily living (ADL) with cuff attachments. We developed and tested a motor-activated wrist orthotic system, which uses a two-tier gesture recognition approach to assist individuals with SCIs to perform a larger range of ADL. The wrist orthotic was easy to learn requiring two or less trials for subjects to perform the gestures once they became familiar with the system. Moreover, there was a low rate of falsely activating the wrist orthotic when performing gestures. The wrist orthotic can be adapted by adding a variety of actuated tools to expand the ability of wearers to perform various ADL.
Introduction
There are approximately 12,500 new cases of Spinal cord injuries (SCI) every year in the United States alone. 53.9% of SCI are in the cervical region (C1-C7) and approximately 44% of these individuals have injuries in the C3-C6 region of the spinal cord (NSCISC, 2014). Daily manual activities such as unlocking doors with keys, swiping credit cards are extremely difficult for individuals with mid-cervical SCIs due to paralysis in the hand muscles preventing grasping and releasing and paralysis or weakness of wrist flexors and extensors (Thomas, Zaidner, Calancie, Broton, & Bigland-Ritchie, 1997).
In order to stabilize a flaccid wrist , wrist orthoses or splints can be used to maintain the normal position of the hand and wrist (Nas, Yazmalar, Şah, Aydın, & Öneş, 2015). Wrist orthotics have often been used in rehabilitation of individuals with SCI to allow for the correct positioning of joints in the wrist, in order to maintain optimal muscle tone and structure. Tenodesis splints can be used for specific tasks such as assisting in picking up small objects by providing support to the thumb and forefinger(Freehafer, 1969; Kang, Park, Lee, & Park, 2013). However, the limited motion of wrist braces for quadriplegics without the ability to flex or extend their wrists principally provide support. With the addition of a pocket in the palm cuff, mid-cervical quadriplegics are able to insert dining utensils, pencils, pens, toothbrushes, or other tools to accomplish certain activities of daily living (ADL) independently. This current research focuses on expanding the functionality and usability of fixed wrist braces.
For individuals with mid-level SCI (i.e. C4-C5), common devices include surface Functional Electrical Stimulation (FES) systems in the form of a forearm sleeve which are applied during early rehabilitation to control voluntary wrist extension for grasping and flexion (Ragnarsson, 2008). Alternatively, several electromechanical exoskeletons have been constructed to provide basic support with hard metal hinges as manipulators (Lucas,et.al, 2004; Orthosis, 2014). Most current systems assist individuals with SCIs through mechanical actuators or ratchet systems activated by existing functional muscles. The drawbacks of these devices are that they are bulky and cause fatigue to the individual (Romilly, Anglin, Gosine, Hershler, & Raschke, 1994). Common ways to control actuators on these systems include speech recognition and gesture recognition (Khokhar, Xiao, & Menon, 2010; Mainardi & Davalli, 2007). Gesture recognition is often achieved through acceleration sensors or electromyography (EMG) signals. Unfortunately, EMG and accelerometer signals by themselves tend to be very noisy and can often lead to false positives (Samadani & Kulic, 2014). While improvements in speech recognition technology provides accurate control of actions during steady state, performance is significantly reduced in noisy environments. Due to the limitations of the aforementioned technologies, a combination of EMG and acceleration or gyroscopic signals have shown to be more accurate and provide better control than through individual application (Georgi, Amma, & Schultz, 2015).
In this paper, we developed and tested the accuracy of a wearable multi-functional wrist orthotic (MFWO) which is activated by EMG signals from the pronator teres (wrist muscle) and concurrently performs distinct functions based on the recognition of four different gestures through an Inertial Measurement Unit (IMU).
Method
Wearable Orthotic
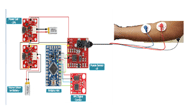
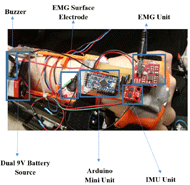
In Figure 1, the wrist orthotic holds an EMG sensor (Advancer Technologies®) with electrodes attached to the surface of the wrist and an IMU (SparkFun Electronic®, which is connected to a microcontroller unit (Arduino® Mini). The MFWO has the ability to incorporate various actuators, such as a servo motor to extend an arm holding a key or card or activating a light or laser pointer. The electronics are powered by two small, rechargeable batteries. The orthotic is designed to be lightweight, weighing less than 200 g which is slightly more than standard commercially available wrist splints.
Gesture Recognition Methods
To minimize the occurrence of false positives, a two-tier gesture recognition approach was implemented to control the MFWO system. The first tier is an EMG sensor which detects a fine gesture allowing the activation of the second tier. The second tier is an IMU which detects one of four gross gestures to perform the desired task.
EMG sensor: System activation
IMU: Gesture Detection
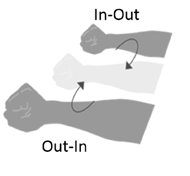
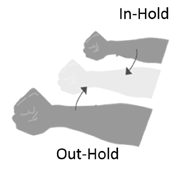
The IMU recognizes four distinct gross gestures: In-Out, Out-In, In-Hold and Out-Hold (Table 1, Figure 2). These gestures were chosen for their comfort and ease of execution by two individuals with cervical SCI. Each of the gestures allows the control of one of the actuators (laser/servo) of the MFWO.
Gesture |
Motion |
In-Out |
Moving hand toward body (In) then away from body (Out) |
Out-In |
Moving hand away from body (Out) then toward body (In) |
In-Hold |
Moving hand toward body (In) and holding position (Hold) |
Out-Hold |
Moving hand away from body (Out) and holding position (Hold) |
Subject Testing
Nine subjects were used to assess the reliability of the gesture recognition system and identify the number of times accidentally activating the EMG sensor (false positives) when wearing the MFWO when performing typical movements. Of these subjects, two were individuals with cervical SCIs and seven were individuals without SCIs.
The subjects were given an unlimited training period to familiarize themselves with the activation of the MFWO and the various gestures. After the training period, they were requested to test the reliability of the system by activating the MFWO system through a double twist of the wrist and forearm and performing each gross gesture five consecutive times. Success was determined when the system correctly and consistently detected the gestures each of the five times. If the system detected the gesture incorrectly, the trial was considered a failure and the subject was asked to restart. The number of trials to achieve success was recorded for each subject. They were then requested to perform eight typical movements to test the reliability in activation of the system and identify false positives (activation of the system with a gesture other than the double twist). These movements include touching one’s forehead, reaching and retrieving an object, grasping, reaching their arms’ inward toward the body, reaching their arms’ outwards away from the body, performing vertical and horizontal circles, and a single sequence of supination-pronation of the wrist (single twist).
Results
System Reliability
Table 2 represents the average number of trials required to successfully complete five consistent correct detections of each gesture. On average each subject required less than 2 trials for the system to correctly perform each gesture command.
| Gesture | Average number of trials for success |
| In-Out | 1.89 |
|---|---|
| Out-In | 1.44 |
| In-Hold | 1.56 |
| Out-Hold | 1.56 |
Table 3 represents the difference in the reliability of individuals with cervical SCI and those without disabilities. The In-Out gesture required the largest number of tries in both, individuals with SCI and those who are able-bodied.
| Gesture | Average number of trials for success | |
|---|---|---|
| With SCI | Able-Bodied | |
| In-Out | 2.5 | 1.71 |
| Out-In | 2 | 1.28 |
| In-Hold | 2 | 1.43 |
| Out-Hold | 2 | 1.43 |
Detection of Accidental Activation
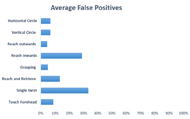
The number of accidental initiating the EMG command (double wrist twist) was determined by how many times the MFWO system was activated through various arm movements of subjects. The activation of the system was identified through a buzzer. As shown in Figure 3, some typical movements activated the system more often than others. It was observed that the three activities that caused the highest false positives were a pronounced, single twist (forearm supination), followed by reaching the arms inwards, and then reaching and retrieving.
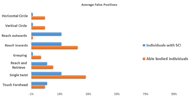
Figure 4 shows the average false positives for individuals with SCI and those who are able bodied. For individuals with SCI, the highest false positives were in the activities of performing a single twist, reaching arms inwards, and reaching their arms outwards. One of the subjects with a C-4/5 SCI was unable to perform the grasping task.
Discussion
On average, the highest false positives were detected for gestures such as performing a pronounced single twist, reaching inwards, and reaching and retrieving. The pronator teres is often activated by either pure wrist flexion or wrist flexion coupled with mild supination (Buchanan, Rovai, & Rymer, 1989), has a rather broad attachment to the elbow and is one of the stronger motors in the forearm (House, Gwathmey, & Lundsgaard, 1976). The pronator teres muscle was chosen for activation of the MFWO in order to provide individuals with upper cervical spinal injuries the ability to control the system with voluntary gross gestures such as the double twist. Other voluntary motor gestures are possible.
Each of the movements which relate to the highest number of false positives has components of either pure flexion or flexion followed by supination in quick succession. However, in individuals with SCI the gesture of reaching outwards resulted in a higher false positive percentage as compared to able-bodied individuals. We hypothesize this is a result of individuals with SCI using a combination of their biceps, forearm muscles, such as the pronator teres, and gravity to extend their arm.
Future Work
Future work will involve the implementation of a machine learning algorithm to improve reliable gesture recognition, further decrease the false positive rate, and allow the system to adapt to individual preferences. Moreover, multiple muscles can be used as aggregated input sources to further validate that a gesture was performed and reduce the false positive rate. This work will also be extended by integrating various tools to the MFWO to assist wearers with different ADL.
References
Buchanan, T. S., Rovai, G. P., & Rymer, W. Z. (1989). Strategies for muscle activation during isometric torque generation at the human elbow. J Neurophysiol, 62(6), 1201–1212.
Center, N. S. C. I. S. (2014). 2014 Annual Report Complete Public Version, 106.
Freehafer, A. A. (1969). Care of the hand in cervical spinal cord injuries. Paraplegia, 7(2), 118–30. http://doi.org/10.1038/sc.1969.22
Georgi, M., Amma, C., & Schultz, T. (2015). Recognizing Hand and Finger Gestures with IMU based Motion and EMG based Muscle Activity Sensing. Proceedings of the International Conference on Bio-Inspired Systems and Signal Processing, 99–108.
House, J. H., Gwathmey, F. W., & Lundsgaard, D. K. (1976). Restoration of strong grasp and lateral pinch in tetraplegia due to cervical spinal cord injury. The Journal of Hand Surgery, 1(2), 152–159.
Kang, Y. S., Park, Y. G., Lee, B. S., & Park, H. S. (2013). Biomechanical evaluation of wrist-driven flexor hinge orthosis in persons with spinal cord injury. J Rehabil Res Dev, 50(8), 1129–1138.
Khokhar, Z. O., Xiao, Z. G., & Menon, C. (2010). Surface EMG pattern recognition for real-time control of a wrist exoskeleton. Biomedical Engineering Online, 9, 41.
Lucas, L., Dicicco, M., & Matsuoka, Y. (2004). An EMG-Controlled Hand Exoskeleton for Natural Pinching. Journal of Robotics and Mechatronics, 16(5), 1–7.
Mainardi, E., & Davalli, A. (2007). Controlling a prosthetic arm with a throat microphone. Conference Proceedings : ... Annual International Conference of the IEEE Engineering in Medicine and Biology Society. IEEE Engineering in Medicine and Biology Society. Annual Conference, 2007, 3035–9.
Nas, K., Yazmalar, L., Şah, V., Aydın, A., & Öneş, K. (2015). Rehabilitation of spinal cord injuries. World Journal of Orthopedics, 6(1), 8–16.
Orthosis, M. H. (2014). ( 19 ) United States.
Ragnarsson, K. T. (2008). Functional electrical stimulation after spinal cord injury: current use, therapeutic effects and future directions. Spinal Cord : The Official Journal of the International Medical Society of Paraplegia, 46(4), 255–274.
Romilly, D. P., Anglin, C., Gosine, R. G., Hershler, C., & Raschke, S. U. A. (1994). Functional Task Analysis and Motion Simulation for the Development of a Powered Upper Limb Orthosis. IEEE Trans, Rehabil. Engng, 2(3), 119–128.
Samadani, A.-A., & Kulic, D. (2014). Hand gesture recognition based on surface electromyography. Conference Proceedings : ... Annual International Conference of the IEEE Engineering in Medicine and Biology Society. IEEE Engineering in Medicine and Biology Society. Annual Conference, 2014, 4196–9.
Thomas, C. K., Zaidner, E. Y., Calancie, B., Broton, J. G., & Bigland-Ritchie, B. R. (1997). Muscle weakness, paralysis, and atrophy after human cervical spinal cord injury. Exp Neurol, 148(0014-4886 (Print)), 414–423.Acknowledgements
We are grateful to the Discovery Park and Center for Paralysis Research at Purdue University for their support. The authors would also like to thank Camila Marrero for her assistance.