Wearable Physiological Telemetry for Individuals with Spinal Cord Injuries to SELF-MONITOR Secondary Health Complications
Shruthi Suresh1, Brendan Raftery2, Bradley S. Duerstock1,3
Weldon School of Biomedical Engineering1, Department of Computer Science2, School of Industrial Engineering3, Purdue University
Abstract
One of the most common post-spinal cord injury (SCI) health conditions is autonomic dysreflexia (AD). Key physiological indicators to identify the onset of AD include changes in pulse and pathological sweating above the SCI level. We developed a self-monitoring physiological telemetry system to help quadriplegics to recognize potential symptoms of AD. Utilizing the commercially-available galvanic skin response (GSR), heart rate, and skin temperature sensors, we developed a mobile computer application to enable wearers to monitor these physiological indicators in real-time and throughout the day. Subjects were recruited to perform typical daily activities, such as typing and using their wheelchairs in order to compare readings between a telemetry system we developed to the new Microsoft® Band™. There was no significant difference between the data being collected by the two systems allowing a transition from the physiological telemetry system to the Microsoft Band.
An implication of this study is to provide long term physiological monitoring for quadriplegics to alert them and their caregivers of the onset of AD.Introduction
Nearly half of all SCIs occur between 16 to 30 years old (NSCISC, 2015). Thus, the years living with disability (YLD) from SCI is among the highest of all diseases and conditions (Polinder et al., 2015; Shavelle, Devivo, Paculdo, Vogel, & Strauss, 2007) 53.9% of SCIs occur in the cervical region (C1-C7) region (NSCISC, 2014). Approximately 30-50% of persons with a SCI are rehospitalized per year due to secondary health complications (NSCISC, 2014). The most common post-SCI health complications are skin pressure sores, autonomic dysreflexia (AD), lack of thermoregulation, deep vein thrombosis (DVT), respiratory complications, urinary tract infections, and orthostatic hypotension (Mayo, 2015; PVA, 2001).
Our goal is to decrease incidences of morbidity and mortality in persons sustaining SCIs by preventing the occurrence of common secondary medical complications associated with SCI, in particular AD. Meticulous physiological monitoring of personal health can reduce the occurrence of SCI secondary complications. However, given the young age of most new SCI patients, there is an understandable lack of diligence in their health monitoring leading to a higher risk of developing post-SCI complications. The mortality rates are significantly higher during the first year after SCI than during subsequent years (NSCISC, 2013).
AD is one of the most life-threatening post-SCI health conditions due to paroxysmal hypertension. AD is caused by hyperactivity of the sympathetic nervous system due to noxious stimulation to the body below the injury site. If this irritation is not relieved, then AD will escalate rapidly resulting in hypertension of over 200 systolic mmHg, which can lead to seizures, stroke or even death. Unfortunately, only 41% of persons with chronic SCI and their family had heard of AD; however, 22% of individuals with SCI reported symptoms consistent with unrecognized AD (McGillivray et al., 2009; PVA, 2001).
During rehabilitation, SCI patients and family members or caregivers are usually educated about how to properly manage AD, including prevention strategies. Most persons with SCI and caregivers can use non-pharmacological methods, such as adjusting body posture and ensuring proper evacuation of the bowel and urinary bladder, to stop AD symptoms. AD symptoms and management varies among individuals. Quadriplegics must learn to become familiar with their own triggers and symptoms (Linsenmeyer, Campagnolo& Chou, 1996). However, in some individuals and instances, it may be difficult to identify what has initiated AD and immediate pharmaceutical medical intervention is required. Moreover, the frequency and severity of AD depends on the individual, some experiencing it on a daily or weekly basis (Elliott and Krassioukov, 2006).
In this study, we developed and tested a wearable physiological telemetry system that enables new quadriplegics to recognize and learn to manage AD episodes they experience. Key physiological indicators of AD include a sudden increase in blood pressure, altered heart rate, sweating above the injury site, facial flushing, and cold and clammy skin (Wan & Krassioukov, 2014) . We had previously validated accuracy of AD measures for galvanic skin response (GSR) through quadriplegic subject testing of sweating during the various activities of daily living (ADL) (Suresh, Duerstock, Duerstock, 2015).
In this study, we compared physiological results that occur when performing typical ADL from a previously developed telemetry system to the MS Band that wirelessly sends measurements related to AD, including pathological sweating, changes in heart rate, and fluctuations in skin temperature, to a mobile tablet for real-time monitoring of potential AD events.
Method
Physiological Monitoring Wristband
 (a)
(a)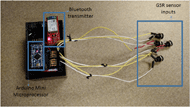
(b)
Mobile Computer Application
A mobile computer application running on Android® operating system was developed to regularly upload physiological data from the MS Band. This application provides easy to read values for HR, GSR measured in Kilo-Ohms resistance, and skin temperature in ⁰F.
Pathological Sweating
In order to detect anomalous changes in GSR activity (pathological sweating) among quadriplegics, normal variations in skin resistance among individual wearers were corrected for during the performance of typical ADL. Baseline GSR measurements above the injury site were determined from a range of male and female quadriplegics with C4-C7 level SCIs (Suresh et al., 2015). In Fig. 1B, the physiological telemetry system can be programmed to alert users when GSR readings exceed normal baseline measurements. Each individual can calibrate the system when performing typical ADL, such as typing on a computer, using a wheelchair for several minutes, and other normal strenuous activities.
Subject Validation
Two quadriplegic subjects with mid-level SCIs, who had participated in baseline GSR measurements, were used to validate the feasibility and reliability of this new telemetry system (Table 1).
| Subject | Level of Injury | Completeness of Injury | Age |
|---|---|---|---|
| Subject 1 | C4/C5 | Complete | 44 |
| Subject 2 | C5/C6 | Complete | 25 |
These subjects re-created similar ADL to the study by Suresh et al. (2015) using both adhesive surface electrodes and the MS Band to measure above the injury GSR data. These activities included relaxing for a duration of two minutes, typing four lines of preset text, playing five games of computerized Tic-Tac-Toe, and continuously travelling in their wheelchairs for five minutes.
Data Normalization
Due to differences in the magnitudes of skin resistance in different subjects, the data was normalized through a min-max method which identifies data in a range of 0 to 1 allowing convenient comparison. The Skin resistance data for each subject SRsub is normalized to SRnorm by subtracting the minimum SRmin and scaling it against the difference of maximum, SRmax and SRmin as shown in Equation (1). SRnorm is referred to as the normalized skin resistance.
(1)
Normalized SR is equal to SRsub minus the minimum value of SR divided by the maximum value of SR minus the minimum value of SR.
Results
Effects of Autonomic Dysreflexia on GSR
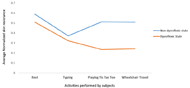
The average normalized skin resistance for Subject 1, who reported AD, progressively decreased during the activities (Fig. 2). However, for Subject 2 a decrease in the average normalized skin resistance was observed during the activity of typing, which was the most physically exhausting ADL.
Comparison of MS Band to Baseline GSR Data
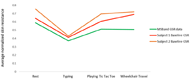
In order to identify the similarity between the GSR data from the MS Band and those identified by the physiological telemetry system developed, a one-way ANOVA test, α= 0.05 was performed on the three distinct sets of data. It was identified that there was no significant difference (p= 0.25) between the GSR data collected by the MS Band and that collected by the physiological telemetry system.
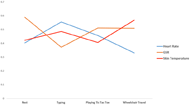
Heart Rate and Skin Temperature
Discussion
Assisting new quadriplegics, where the most frequent age of sustain a SCI is 19 years old (NSCISA, 2013), to be more cognizant of potentially dangerous secondary health complications could significantly reduce the incidence of rehospitalization, decrease healthcare costs, and be potentially life-saving. Unfortunately, due to the foreign reactions of one’s body to noxious stimuli to paralyzed parts of the body, it can be difficult for quadriplegics to elucidate what may be occurring in their bodies. AD, which is caused by hyperactivity of the sympathetic nervous system, can be particularly difficult to isolate the source of noxious stimuli, such as a full bladder or pressure sore. Through the development of the wearable telemetry system, we aim to allow individuals with SCI greater self-sufficiency and safety, which is critical to helping persons with SCIs to successfully return to their communities.
Effects of AD on the values of GSR and Skin Temperature
One of the most common physiological indicators of AD is sweating above the injury site (Wan & Krassioukov, 2014). Since sweating causes a decrease in skin resistance, an event of AD will result in decrease in the average normalized skin resistance, as shown in Figure 2. Moreover, as discussed in Suresh, et al (2015) this result is in agreement with the findings of other researchers in the field wherein they identified a reduction in GSR in activities that cause higher stress or exertion.
Another common indicator of AD is cold and clammy skin (Krassioukov AV, et al., 2003). This sensation was reported by the subject who felt dysreflexic based on previous experience. There was a positive correlation between GSR and skin temperature for this subject. As GSR decreased with sweating, there was a decrease in skin temperature. However, more data is required as there was no overall significant difference identified between GSR and temperature measurements.
Comparison of MS Band to Baseline GSR Data
The comparison of GSR data acquired from the MS Band as well as the Physiological Telemetry system shows no significant difference between the data sets. Thus, the MS Band seems to perform as well as the Physiological Telemetry system that we had previously developed with dedicated sensors control through an Arduino® microcontroller. Therefore, future work to evaluate a wearable telemetry system may use the MS Band, which is compact and comfortable to wear for long periods of time.
Heart Rate and Exertion Levels
For the subject who did not experience AD, there was an inverse relationship between heart rate and GSR. This supports that as GSR decreases with increased exertion, the heart rate increases. However, in activities where lesser exertion was reported, the heart rate decreases (Bhambani, 2002).
Future Work
Future work will involve using the MS Band to collect baseline GSR, Heart rate and skin temperature data of subjects with SCI during their daily lives. This will allow the development of a telemetry system which can be used to detect any physiological changes from the pre-determined baseline. More subjects will also be recruited to develop a comprehensive representation of the different parameters to develop an accurate and reliable system.References
Elliott, S., & Krassioukov, A. (2006). Malignant autonomic dysreflexia in spinal cord injured men. Spinal Cord, 44(6), 386-392.
Karlsson A (1999) Autonomic dysreflexia. Spinal cord 37:383-391
Krassioukov AV, Furlan JC, Fehlings MG (2003) Autonomic dysreflexia in acute spinal cord injury: an under-recognized clinical entity. Journal of neurotrauma 20:707-716
Linsenmeyer, T. A., Campagnolo, D. I., & Chou, I. H. (1996). Silent autonomic dysreflexia during voiding in men with spinal cord injuries. The Journal of urology, 155(2), 519-522.
Mayo Clinic website, Health information (2015). Retrieved: September 21, 2015 http://www.mayoclinic.org/diseases-conditions/spinal-cord-injury/basics/complications/con-20023837
McGillivray, C. F., Hitzig, S. L., Craven, B. C., Tonack, M. I., & Krassioukov, A. V. (2009). Evaluating knowledge of autonomic dysreflexia among individuals with spinal cord injury and their families. The journal of spinal cord medicine, 32(1), 54.
National Spinal Cord Injury Center (2015). Annual Report for the Model Spinal Cord Injury Care Systems.
National Spinal Cord Injury Center (2013). Annual Report for the Model Spinal Cord Injury Care Systems 2013.
Polinder, Suzanne, Meerding, Willem Jan, Mulder, Saakje, Petridou, Eleni, & van Beeck, Ed. (2007). Assessing the burden of injury in six European countries. Bulletin of the World Health Organization, 85(1), 27-34.
Paralyzed Veterans of America (2001). Acute management of autonomic dysreflexia: individuals with spina cord injury presenting to health-care facilities Washington, DC.
Shavelle RM, Devivo MJ, Paculdo DR, Vogel LC, Strauss DJ. (2007). Long-term survival after childhood spinal cord injury. J Spinal Cord Med. 2007;30 Suppl 1:S48-54.
Suresh, S., Duerstock, H., & Duerstock, B. (2015). Skin Resistance as a Physiological Indicator for Quadriplegics with Spinal Cord Injuries During Activities of Daily Living. In Smart Health (pp. 157-168). Springer International Publishing.
Wan, D., & Krassioukov, A. V. (2014). Life-threatening outcomes associated with autonomic dysreflexia: A clinical review. The Journal of Spinal Cord Medicine.Acknowledgements
This project was supported by the Indiana Spinal Cord and Brain Injury Research Fund through the Indiana State Department of Health (awarded to B.S.D.). We are grateful to the Discovery Park and Center for Paralysis Research at Purdue University for making this project possible. We would also like to thank Microsoft Research and Dr. Arjmand Samuel for access to Microsoft technologies during this project