In this study, a surface electromyographic (SEMG) is used to identify four hand motions_reach to grasp, back to release, supinate to palm up, pronate to palm down. From the analysis of the single-chip-based (Intel89C51) control system, a one channel SEMG is used to distinguish four hand motions for the short below elbow amputee. One surface electrode is placed on the biceps brachii. The Intel89C51 microprocessor integrates the signal processing module. The on-line controller can provide 95% correct rate for the discrimination of four hand motions.
BACKGROUND
The myoelectrically controlled prostheses provide amputees extra prosthetic options. However, most commercial prosthetic hands (e.g. Steeper Electric Hand, Otto Bock System Electric Hand, Swedish Systemteknid Hand) are one degree of freedom grippers that are controlled by one or two channel of SEMG signals. Otherwise, proportional myoelectric control has been used in the Utah artificial arm. Those SEMG controlled systems mentioned above limit the ability of manipulation and the complexity of SEMG patterns recognition [1-3].
In this study, we proposed a simplified and alternative one channel SEMG controlled prosthetic hand. The one channel SEMG was used to drive tow motors. One of the motors was responsible for reaching to grasp or backing to release. Another motor was used to supinate or pronate.
METHOD
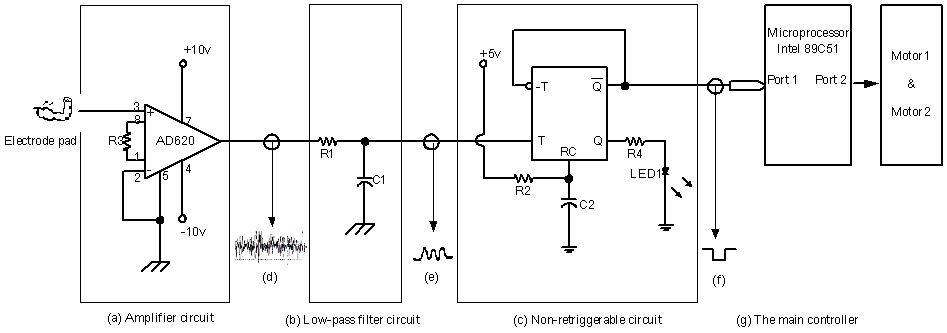
The circuit diagram of prosthetic hand controlled using SEMG, as shown in Figure 1. The circuit includes an amplifier circuit, a filter circuit, a non-retriggerable circuit, etc. The electrode (developed B&L Engineering motion lab.) with a pre-amplifier applied to the stable muscle groups (e.g. the related muscle groups of the upper extremity) of an operator. The SEMG signal from the patient's movement can be obtained through one instrumentation amplifier circuit-AD620, gain: 500, as shown in Figure 1 (a) and one low pass filter, cut-off frequency=20Hz, as shown in Figure 1 (b). The signal is obtained when the patient's muscle groups are active (flexed). Because the SEMG signal is a complicated signal that changes rapidly with time and is difficult to serve as the control input signal, a non-retriggerable circuit was designed to assure that a stable pulse is transferred to the controller after the user stimulates the muscle once. This non-retriggerable circuit, as shown in Figure 1(c), is modulated through R1 and C1 using formula (1).
τ= 0.453 * R1 * C1 ........ (1)
The various non-retriggerable periods (τ) can be set. A stable pulse can be output to the main controller when the operator's muscle is activated (flex-relax) once. The input and output waveforms of the SEMG signal processing circuit are shown in Figures 1(d), 1(e) and 1(f). Because patients may generate SEMG signals at different intensities, a reference voltage regulator for controlling the SEMG signal was added to the amplifier as a part of this circuit design. Each patient can set up a reference point defined as the SEMG signal when the muscle is relaxed. Each time after the operator's muscle flexes-relaxes once a pulse will be output to the main controller.
The main controller adopted
in this research was the Intel 89C51 microprocessor. The
Intel 89C51 microprocessor has four I/O ports (Port 0,
Port 1, Port 2 and Port 3) and each I/O port has 8 bi-directional
bits. Port 1 of this microprocessor is used to receive
the control signal output by the SEMG processing circuit,
for selecting the hand motion functions. Port 2 can be
used to drive the motor 1 for reaching to grasp or backing
to release while it received a pulse
( ![]() ).
Also, Port 2 can be used to drive the motor 2 for supinating
to palm up or pronating to palm down while it received
two pulses continuously (
).
Also, Port 2 can be used to drive the motor 2 for supinating
to palm up or pronating to palm down while it received
two pulses continuously ( 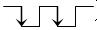 ).
).
 |
RESULTS
The system software implementation and hardware design are completed. This includes designs such as the SEMG input (as shown in Figure 2) and the software for the single mouse-scanning window. The SEMG input now is modularized, it could be implemented into any home appliances. The disabled only have to attach the SEMG electrode to the appropriate muscle to operate this system. For example, an operator only has to place the electrode with the pre-amplifier onto his biceps brachii or other related muscle group. When the muscle is active (flexed once), the SEMG signal can be used to control the prosthetic hand.
Eleven volunteers were recruited in this system assessment. The average age of these volunteers was 41.3±5.7. The subjects had amputated or a similar hand dysfunction and had been discharged from the hospital over 6 months. The subjects exhibited no muscle spasms and were able to use a personal computer before their injuries. The electrodes were placed on their biceps brachii muscles. After 30 minutes' training, the clinician gave each subject 10 command movements (the right upper limb flexed and then relaxed immediately). The average accuracy of trigger pulse signal generated by this device was 95.5±7.2%.
 |
 |
DISCUSSION
Based on the results from the system assessment, the SEMG input functions effectively. The 5% inaccuracy did not result from the system function, but from operator inexperience with the system. Enhancing patient training can lower the performance inaccuracy.
We hope that this circuit design can become an efficient instrument for the disabled to overcome daily inconveniences. The purpose of this research was to eliminate using a complexity instruments (SEMG patterns recognition) with inconvenience or high cost. Direct SEMG input through remaining muscle functions has been demonstrated a viable alternative to more expensive methods. Through this system, the disabled can have the opportunity to win back their dignity and lead independent lives.
REFERENCES
ACKNOWLEDGMENTS
This work was supported by the National Science Council, ROC, under Grants NSC 91-2614-E-146-001.
Yu-Luen Chen,
Ph.D., Associate Professor & Chairma
Department of Electronic Engineering, Hwa-Hsia College
of Technology and Commerce
No.111, Hwa-Shin St., 235
Chung-Ho, TAIPEI, TAIWAN, ROC
886-2-89415158
886-2-23613331 (fax)
allen@cc.hwh.edu.tw
