ABSTRACT
The tightness of an orthotic brace varies greatly during daily activities. If treatment efficiency depends on how tight the patient wears his/her brace, the brace should be adjusted to the prescribed tightness level regularly. Subjective feeling is the most commonly used method to evaluate how patients tighten their braces. To provide an objective measure, a battery-powered microcomputer system was developed to monitor loads exerted by braces during daily living. The system not only records how well the brace has been used, but also helps patients wear the brace worn as prescribed. This system has been used by scoliotic patients and feedbacks from them suggests the system is very easy to use and helpful.
BACKGROUND
There are many orthotic braces on the market. A common brace to treat scoliosis is a plastic shell that provides mechanical support to the spine (1) when moderate curvature of the spine is detected. It is suggested that to be effective, these braces must be worn for up to 23 hours per day until the child has completed skeletal growth. Successful brace treatment is also thought to require patients to wear braces as prescribed including securing the straps to provide a level of pressure on the convexity of the curve. The concerns from orthopaedic surgeons are: do patients wear the brace as prescribed and is the correction due to mechanical support.
The commonly accepted methods to assess compliance are to ask the family if the brace is being used and to visually inspect the brace for signs of wear. To investigate how tight patients wear their braces during their normal usage, patients at the scoliosis clinics are asked to demonstrate how they tighten their braces. A mark is usually placed on the brace straps to help properly tighten the braces. However, normal growth and stretching of the brace straps occurs causing the mark to be inaccurate. Most braces have an opening and straps at the back which makes it difficult for the brace wearer to see the strap marks. Other methods have been used to study how loads are applied while patients wear their braces (2,3). However, these two studies were conducted in laboratory settings, which may not be true indicators of the pressure exerted by braces during daily activity over a treatment period lasting typically 2 years. Therefore, a low-powered portable device was required to monitor the loads exerted by braces used to treat children with spinal deformities during daily activity. This system should also be able to monitor the tightness of other orthotic braces.
Research Question
The objectives of this study were to evaluate the feasibility of the approach and to determine the correlation between brace treatment with the tightness and compliance of brace wear.
METHOD
The monitoring device consists of a programmable digital data acquisition system and a force transducer (FS01, SenSym Inc.). The operating force range of the transducer is 0 - 6.82N. This transducer was selected mainly because of its low profile and force range. The monitoring system measured and recorded the time and temporal profile of the loads on the pressure pad imposed on the trunk during daily activity. Although this study included subjects with Boston braces only, the system could be used for other orthotics and prosthetics devices. Also, any other force transducer that could operate in a +5V power supply and its force range within the specific applications could be chosen. Figure 1 shows a force sensor and a Boston brace. The force transducer was placed within the pressure pad that was intended to apply the largest force loads to the body.
The low-powered load-monitoring device was packaged into a plastic box with dimensions 5.5cm x 8cm x1.7cm (Figure 2) and weight of 60g. Three light emitted diodes (LEDs) were used to indicate the tightness level. The label H (high load) LED (red) signifies a tightness above 120% of the prescribed level; while the middle (accepted load) LED (green) signifies tightness within 80 to 120% of prescribed; the L (low load) LED (red) signifies tightness below 80% of prescribed. The prescribed tightness was set by the physician or orthotist after the force transducer was installed. This feature helps the subjects wear their braces at the prescribed tightness. The system was powered by two AAA-sized Nickel-Metal-Hydride (Ni- MH) rechargeable batteries. The subject was required to recharge the microcontroller every day for 2 hours when she was not wearing her brace.
 |
 |
RESULTS
Eighteen brace candidates, 3 males and 15 females age 13.6 + 1.8 years, who have worn their braces from 6 months up to 1 year were recruited. All subjects gave their informed consent to participate in this study. The subjects used the device to measure the daily force patterns exerted by the brace upon the trunk. The average prescribed force was 1.33 ± 0.65 N, and the prescribed brace time was 21.1 ± 2.77 hours/day. The force level was zero when the brace was not worn. The brace was not used approximately 23 ± 20% of the total time. The percentages of the total time that the force level was below 80%, between 80 to 120%, and above 120% of the prescribed level were 24 ± 15%, 14 ± 16% and 12 ± 11%, respectively. Approximately 27 ± 25% of the total time the device did not collect data. The major reason was due to the patients did not recharge the system regularly.
Only 12 subjects completed their brace treatment. The duration of the brace treatment was those subjects were 24.6 + 4.5 months. The time at which they used the monitoring device was 8.2 + 3.6 months after they were first prescribed their braces. One subject had curve improvement, 7 subjects had no change and 4 subjects had curve deterioration. Figure 3 summarizes the quantity and quality of how well braces were worn with the treatment outcomes. The improvement subject was 84% compliant and wore her brace above or in the target load range 62% of prescribed time. No change subjects were 70% compliant and wore brace above or in the target load range 40% of prescribed time. Deterioration subjects were 64% compliant and wore brace above or in the target load range only 26% of prescribed time.
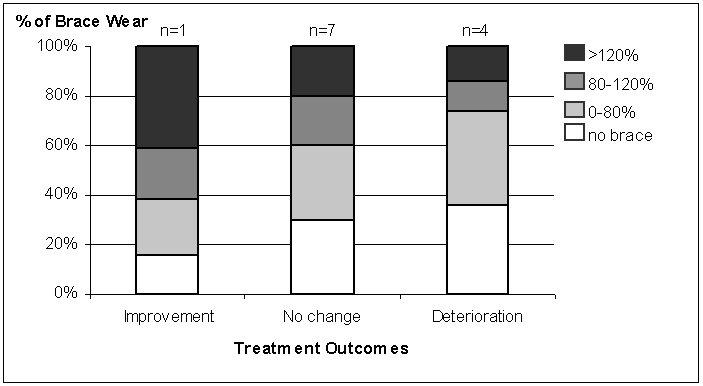 |
DISCUSSION
The main purpose of brace treatment is to cease curve progression during the high-risk growth period of the adolescent growth phase. Any reduction in the curve or deformity due to bracing is usually considered a "bonus". It appears that tightening the straps to the prescribed level and wears the brace as much as the prescribed time is important for the brace treatment. Simply wearing a brace is not sufficient to neither effect an improvement in spinal curvature nor prevent collapse.
REFERENCES
-
Asher MA, Whitney WH.: `Orthotics for Spinal Deformity', Orthotics Etcetera, Baltimore, 1986.
- Jiang H, Raso VJ, Hill DL, Durdle NG, Moreau M (1992). Interface Pressures in the Boston Brace Treatment for Scoliosis. Proceedings Int'l Symposium on 3-D Scoliotic Deformities, Montreal, 27-30 June, p395-399.
-
Wong MS, Mak AF, Luk KD, Evans JH, Brown B (2000). Effectiveness and biomechanics of spinal orthoses in the treatment of adolescent idiopathic scoliosis (AIS). Prosthetics & Orthotics International 24, 148-162.
Edmond Lou, Ph.D.,
P.Eng
Glenrose Rehabilitation Hospital
10230 - 111 Ave Edmonton, AB, Canada T5G 0B7
(780) 471-2262 ext 2688
email: Edmond_lou@shaw.ca
