1Callaghan Innovation, Christchurch, New Zealand;
2Burwood Academy of Independent Living, Christchurch, New Zealand
ABSTRACT
Seven chronic stroke survivors (5 female) aged 59.4 – 79.6 years completed a 4 - 6 week upper limb training program using a table-supported computer input device (the Able Reach) to play bespoke computer games designed to encourage a large number of goal oriented arm movements. Over the course of the intervention, participants received between 9 (n = 4) and 16 (n = 3) hours of game play. On average, Fugl-Meyer scores increased by 4.9 over the course of the intervention, ranging up to 9 points. One participant gradually deteriorated throughout the trial. These results suggest that the Able Reach is a useful adjunct to regular physical therapy in a stroke population.
BACKGROUND
Stroke is a leading cause of disability in the developed world, with as many as 20% of survivors being unable to use their upper-limb and as few as 5% achieving full recovery of the upper-limb (Nakayama et al 1994). It is likely that this poor recovery is due at least in part to insufficient rehabilitation. The mechanism underlying motor learning in general, as well as motor relearning post stroke, is neural plasticity (Kleim and Jones 2008). Neural plasticity is the central nervous system’s ability to adapt by altering its structure and/or function in response to changing internal and external pressures. Animal data suggest that between 400 and 600 repetitions per session are required to elicit the neuroplastic change that underpins functional recovery (Kleim et al 1998; Nudo and Milliken, 1996). However, it is likely that standard care currently provided to most stroke survivors falls far short of this, with one study reporting as few as 2 – 39 repetitions per session of a given activity during upper limb therapy sessions for people recovering from (Lang et al 2007).
In contrast to conventional therapy, robots provide a means of delivering high dosage therapy, with hundreds of repetitions of goal oriented movements carried out in every therapy session (Huang and Krakauer 2009). While robotic rehabilitation shows great potential for facilitating recovery from stroke (Huang and Krakauer 2009) existing robots are prohibitively expensive and complex for most clinical and home settings. Inspired by the success of robotic rehabilitation, we have developed a low-cost system of rehabilitation devices which draws on the concepts of robotic rehabilitation devices. Our system includes purpose built computer games which encourage a large number of point to point reaching movements. We have shown previously that the games are highly motivating and participants enjoy playing them (Sampson et al 2012; Hijmans et al 2011; King et al. 2012). The system comprises individual devices that align to the degree of impairment presented. This paper reports on the preliminary results of a clinical trial using one of these devices – the Able Reach – which targets stroke survivors with moderate upper limb impairment.
METHODS
The Able Reach and Games
The Able Reach is a unilateral, gravity supported exercise device which is operated on a table-top, similar conceptually to traditional sliding board exercises (Fig 1). It was first described in King et al. (2010) and further developed now into a tool suitable for trial. The hemiparetic arm is strapped onto the Able Reach which controls the mouse cursor on a computer screen. The Able Reach is used in conjunction with a suite of computer games designed specifically for stroke survivors, which are clear and easy to see and provide a graduated series of physical challenges, from stationary target hitting, to strategic target hitting to moving target hitting games. The magnitude of Able Reach movement, and corresponding arm movements, required to navigate across the computer monitor is adjustable to suit a wide range of abilities and to ensure the participant is continually challenged as their reach ability improves.
Participants
Seven post-stroke participants with upper limb hemiparesis have been recruited to date. The mean age of the participants was 69.7 ± 8.3 years (range 59.4 – 79.6 years). The mean time post stroke at the time of first assessment was 15.2 ± 15.0 months (range 5.9 – 48 months).
Assessment, intervention and outcome measures
All participants provided their informed consent and this study was approved by the New Zealand Health and Disabilities, Upper South A, Regional Ethic Committee. The intervention consisted of a 4 – 6 week period where participants played computer games using the Able Reach in their own homes, generally using their kitchen table as the support surface. The participants had a graded introduction to the games, initially starting with the easiest (stationary target hitting with large targets and no time pressure) until they were accustomed to the set up. As participants became more familiar with the system, more challenges were introduced (smaller targets, moving targets, time pressure etc.). A typical first session would last for 20 – 30 minutes and was incremented across the course of the first week until participants were able to complete 45 – 60 min of game play in each session. Sessions were supervised by one of the three authors who ensured that the participant played a variety of games, focused on producing quality movement, and that game challenge was adjusted appropriately (increased or decreased as necessary). Participants 1 – 4 received approximately 9 hours of game play (3 x 45-min sessions per week for 4 weeks); the protocol was adjusted to accommodate the findings of Kwakkel et al (2004) for the last 3 participants such that they received at least 16 hours of training (4 x 45-min sessions per week for 6 weeks until 16 hours had been achieved).
The Fugl-Meyer upper limb motor function test (FMA-UL) was the primary outcome measure used, which was provided by one of the authors (MS), a registered physiotherapist. Participant motivation was also assessed using the Intrinsic Motivation Inventory (IMI), a 32 question form which quantifies the subjective experience of a participant resulting from the intervention. It assesses 5 categories of experiences – Interest/Enjoyment; Perceived competence; Effort/Importance; Perceived choice; and Value/Usefulness. In total, participants were assessed using the FMA-UL 4 times – upon enrolment in the study (T0), 4 weeks later, during which time no intervention was supplied (T1), post- intervention (T2), and finally 4 weeks after the intervention was completed (T3). The intervention started within a week of T1; T2 was carried out within a week of the intervention being completed. The IMI was applied at T2 for 6 out of the 7 participants, the remaining participant did not complete the IMI.
Any existing recovery was established by T0 and T1 - if the FMA-UL score differed by 2 points or less between T0 and T1, the participant recovery was regarded as having plateaued. This was the case for 6 out of the 7 participants, with the remaining participant having an increase of 3 points. Any difference in FMA-UL score between T1 and T2 can therefore be primarily attributed to the effect of the intervention. The difference between T2 and T3 indicated how well any improvements in function were maintained post therapy.
A repeated measures ANOVA was used to test for differences in FMA-UL score across the 4 assessments and post-hoc analysis was done using the Bonferroni correction.
RESULTS
Figure 2a illustrates the change in FMA-UL across the 4 assessments for each participant, with the group average change in Figure 2b. There was a significant effect for assessment on the FMA-UL score (p = 0.006), with post hoc analysis revealing that there was no difference between T0 and T1 (p = 1); FMA-UL increased significantly from T1 to T2 (p = 0.004); FMA-UL was greater at T3 than T1 (p = 0.01) and lastly there was no difference in FMA-UL between T2 and T3 (p = 1). The average increase in FMA- ULfromT1-T2was4.9,andfromT1-T3was4.3.Table 1 shows the IMI scores (as percentages) for the 6 participants who completed it.
DISCUSSION
| Participant | Interest/ Enjoyment |
Perceived |
Effort/ |
Perceived |
Value/ |
Average |
|---|---|---|---|---|---|---|
| 1 | ||||||
| 2 | 98 | 76 | 97 | 98 | 100 | 94 |
| 3 | 82 | 64 | 83 | 88 | 94 | 82 |
| 4 | 96 | 95 | 100 | 100 | 98 | 98 |
| 5 | 59 | 64 | 66 | 49 | 88 | 65 |
| 6 | 84 | 83 | 80 | 65 | 73 | 77 |
| 7 | 100 | 90 | 94 | 100 | 100 | 97 |
| Average | 86 | 79 | 87 | 83 | 92 | 86 |
A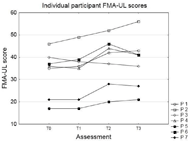 B
B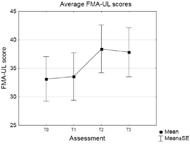 Figure 2: Individual (A) and group average (B) Fugl-
Figure 2: Individual (A) and group average (B) Fugl-
Meyer Assessment Upper-Limb scores for all four
assessments.
Rehabilitation is primarily a process of relearning how to move (Carr and Shepherd 1987), and as with learning any skill, recovery from stroke is maximized by performing a large number of repetitions of goal directed movements. We estimate that participants in this study performed between 500 and 800 goal directed reaching movements per 45 minute session. The results show an average increase of 4.9 points on the FMA-UL as a result of a 4 – 6 week intervention using the Able Reach and computer games. This increase falls within the range of estimated clinically important differences for the FMA-UL in chronic stroke reported by Page et al (2012) and was maintained for 4 weeks post-intervention where no additional training was given. On average, the participants who received 9 hours of therapy had an increase of 4.3 points on the FMA-UL scale between T1 and T2, while the participants who received 16 hours of therapy had an average increase of 6 points. At the final assessment however, the average increase from T1 was 4.5 for the 9 hour group and 4 for the 16 hour group. Our sample size is too small to draw any meaningful conclusion from this observation, but it is interesting to note that there are no obvious gains at this stage from the extra hours of therapy.
Our results compare very favourably with robotic interventions for upper limb rehabilitation in chronic stroke populations. For example, Fasoli et al (2003) trialled two different robotic interventions and reported increases of 3.2 and 3.7 FMA-UL points above baseline measures. Similarly, Lo et al (2010) compared robotic rehabilitation to standard care and to intensive therapy. FMA-UL scores increased by 1.1 and 3.9 points compared to baseline for both robotic rehabilitation groups.
In general, improved FMA-UL scores were observed for all except one participant, with increases ranging from 3 to 9 points between T1 and T2. The exception to this was participant 3, who although healthy enough to maintain participation in this trial, unfortunately experienced a noticeable decline in overall health across the course of the trial (due to a leg ulcer) which may have contributed to a lack of observed benefit of our intervention. It is also noteworthy that participant 2 had a steady increase in FMA- UL from T0 – T3, suggesting that our intervention may not have been a significant factor in their recovery. While all of the participants in this study were chronic stroke survivors, the group was heterogeneous, with initial FMA-UL scores ranging from 17 to 46 across participants. It is therefore not surprising that not all participants demonstrated a clear benefit from using the Able Reach. When we consider only the FMA-UL scores of the 5 participants who did demonstrate a clear benefit, the average increase between T1 and T2 is 6.4 points, and between T1 and T3 is 5 points. The average (across all categories) score on the IMI ranged from 65 to 99% across participants, with the overall average score being 86%. This indicates that in general participant were highly motivated by the intervention and found it to be both enjoyable and useful. The participants were monitored closely for negative side effects, such as arm pain, that may have resulted from the intervention. One participant reported slight arm stiffness after the initial few sessions, one other reported shoulder pain at T1 which had resolved by T2. Other than this, no pain was reported by any of the participants as a result of Able Reach use, suggesting the device was well tolerated by the participants.
CONCLUSION
Our results provide evidence that the Able Reach in conjunction with bespoke computer games can significantly reduce upper limb impairment in chronic stroke survivors, is well tolerated and found to be motivating, useful and enjoyable. Future research will include a larger clinical trial to confirm these results as well as automating the system so that it can be used without direct supervision.
REFERENCES
Carr, J. H., & Shepherd, R. B. (Eds.) (1987). Movement Science: Foundations for Physical Therapy in Rehabilitation. Rockville, MD: Aspen.
Fasoli, S. E., Krebs, H. I., Stein, J., Frontera, W. R., & Hogan, N. (2003). Effects of robotic therapy on motor impairment and recovery in chronic stroke. Archives of Physical Medicine and Rehabilitation, 84, 477-482.
Hijmans, J. M., Hale, L. A., Satherley, J. A., McMilan N. J., & King, M. J. (2011). Bilateral upper limb rehabilitation after stroke using a movement based game controller. Journal of Rehabilitation Research and Development, 48, 1005 – 1013.
Huang, V. S., & Krakauer, J. W. (2009). Robotic neurorehabilitation: a computational motor learning perspective. Journal of NeuroEngineering and Rehabilitation, 6, article no. 5.
King, M., Hale, L., Pekkari, A., Persson, M., Gregorsson, M., & Nilsson, M. (2010). An affordable, computerized, table-based exercise system for stroke survivors. Disability and Rehabilitation Assistive Technology, 5 288-293.
King, M. J., Hijmans, J. M., Sampson, M., Satherley, J., & Hale, L. (2012). Home-based stroke rehabilitation using computer gaming. New Zealand Journal of Physiotherapy, 40, 128-134.
Kleim, J. A., Barbay, S., & Nudo, R. J. (1998). Functional reorganization of the rat motor cortex following motor skill learning. Journal of Neurophysiology, 80, 3321- 3325.
Kleim, J. A., & Jones, T. A. (2008). Principles of experience-dependent neural plasticity: Implications for rehabilitation after brain damage. Journal of Speech, Language, and Hearing Research, 51, S225-S239.
Kwakkel, G., van Peppen, R., Wagenaar, R. C., et al. (2004). Effects of augmented exercise therapy time after stroke: a meta-analysis. Stroke, 35, 2529-2539.
Lang, C. E., MacDonald, J. R., & Gnip C. (2007). Counting repetitions: An observational study of outpatient therapy for people with hemiparesis post-stroke. Journal of Neurologic Physical Therapy, 31, 3-10.
Lo, A. C., Guarino, P. D., Richards, L. G., et al. (2010). Robot-assisted therapy for long-term upper-limb impairment after stroke. The New England Journal of Medicine, 362, 1772-1783.
Nakayama, H., Jorgensen, H. S., Raaschou, H. O., & Olsen, T. S. (1994). Recovery of upper extremity function in stroke patients: the Copenhagen Stroke Study. Archives of Physical Medicine and Rehabilitation, 75, 394-398.
Nudo, R. J., & Milliken, G. W. (1996). Reorganization of movement representations in primary motor cortex following focal ischemic infarcts in adult squirrel monkeys. Journal of Neurophysiology, 75, 2144-2149.
Page, S. J., Fulk, G., D., & Boyne, P. (2012). Clinically important differences for the upper-extremity Fugl- Meyer scale in people with minimal to moderate impairment due to chronic stroke. Physical Therapy, 92, 791-798.
Sampson, M., Shau, Y. W., & King, M. J. (2012). Bilateral upper limb trainer with virtual reality for post-stroke rehabilitation: case series report. Disability and Rehabilitation: Assistive Technology, 7, 55-62.
ACKNOWLEDGEMENTS
We thank the participants for willingly giving their time to our research and opening their homes to us.
